Enzymes are hugely important in the human body both for normal function and for drug therapies. Their kinetics (or function with respect to time) are also easily graphable, making them a very testable MCAT topic. Even more importantly, once you’ve aced the big, bad MCAT, you will need to revisit enzyme kinetics in every medical school curriculum, so building a good foundation now will serve you well later!
Below is a “cheat-sheet” to accompany a more in-depth review of enzyme kinetics using review books and/or online videos. It summarizes the important concepts you need to understand BEFORE you get to Michaelis-Menten kinetics, Lineweaver-Burke plots, and understanding various types of inhibition.
Review of enzymes:
Enzymes
- Enzymes are proteins that interact with molecules (substrates) in the body to help transform them into other forms or combine them with other molecules (products)
- Enzymes are reusable; a single enzyme performs the same reaction many, many times
- Enzymes have an active site where they interact with specific substrates (the shape and chemical properties of the active site determine which substrates it will interact with and how readily it will interact with each one)
- Active sites are enzyme- and substrate-specific
Theories of enzyme-substrate binding
- Lock and Key Model: substrates fit into active sites that are optimally shaped for these substrates like a key fits into a lock
- Induced Fit Model: a substrate kind of fits into an enzyme’s active site; the unoccupied binding site has some affinity for the substrate when it’s empty, but when the substrate comes close enough, the binding site undergoes a conformational change that makes it fit the substrate perfectly. Think about this like putting your hand into a glove. When you are holding the glove in your hand, it looks kind of like a hand, but when you put your hand INTO the glove, it fits very snugly against your skin.
Reversible reactions
- In every reversible reaction, A ↔ B, you have molecule undergoing the reaction in both directions, at a rate of k1 (A → B) and k2 (B → A)
- If k1 > k2, the reaction will favor the A → B direction, and you will end up with a larger concentration of B than A at equilibrium
- Equilibrium: the proportions of A and B such that the concentrations of A and B remain the same
- The substrate can turn into the product and the product can turn back into the substrate (remember Le Chatelier’s principle!). When a reaction is at equilibrium,that means that it has reached the balance of substrate and product concentrations that makes it “happy.” The NET reaction does not proceed in either direction. IMPORTANT: at equilibrium, the forward and reverse reactions are still continuing, but at the same rate, so there is no NET change in product vs. substrate concentration; in other words, the net reaction produces no change in the proportion of A vs. B
Enzyme catalysis
- A and B both have a certain amount of energy, and in order to get from A to B you have to go through a transition state, which requires a lot of energy to reach. The energy required to reach the transition state is the activation energy required to get the reaction to “go”
- Enzymes are CATALYSTS, which means that they lower the activation energy for a given reaction, thereby increasing the reaction rate
- Enzymes do not affect the equilibrium of a reaction (adding an enzyme will not shift the equilibrium concentration to suddenly favor more A than B if the natural equilibrium of the reaction is to favor more B than A)
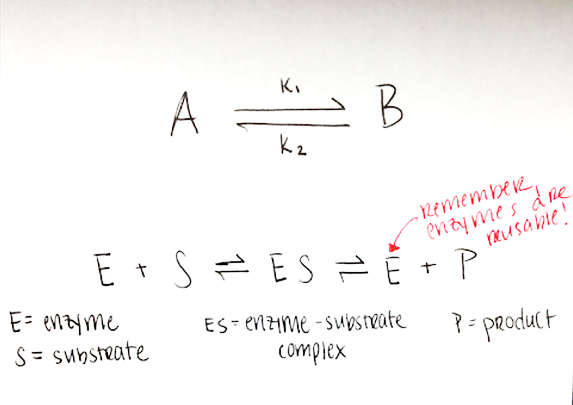
Review of key terms:
V = reaction rate → the amount of product formed per unit time; depends on:
- Reaction conditions (in the body, some reactions take place optimally at certain temperatures, pHs, etc.)
- Substrate concentration = [S]
- Imagine a bakery making crème brulee. One person makes the custard (substrate) and one person fires it with a blowtorch (enzyme). If there are more custards prepared, you will be able to use the blowtorch to make more finished crème brulees.
- Enzyme concentration = [E]
- Adding an enzyme will increase the reaction RATE by lowering the activation energy, but some reactions will occur without the help of an enzyme (more slowly)
- If some of your blowtorches aren’t working (in the body this could happen from inhibition of function by certain drugs), you will make fewer crème brulees per unit time.
- Some reactions will NOT proceed without an enzyme because the activation energy is too high in the absence of an enzyme
- In the presence of an enzyme, the enzyme’s affinity for the substrate (Km, discussed below) can also affect the reaction rate
V0 = initial velocity = initial rate of reaction
- This is the rate of reaction when you first add a little bit of substrate into your beaker full of enzyme.
- As you accumulate more and more of your product, you approach the equilibrium of the reaction, so your reaction velocity will slow down. This is because with higher concentrations of product, more individual molecules of product are spontaneously undergoing the reverse reaction. The reaction is still progressing in the NET forward direction, but the overall rate of product accumulation is slowing since you lose some product to the reverse reaction.
Vmax = maximum reaction rate
- Specific to each enzyme and reaction
- No matter how much substrate you have, a single enzyme has a maximum capacity for completing the reaction (thinking about the blowtorch example, a single blowtorch, no matter how much gas it has supplying it or how many crème brulees are waiting to be fired, has a limit to how quickly it can fire a single crème brulee)
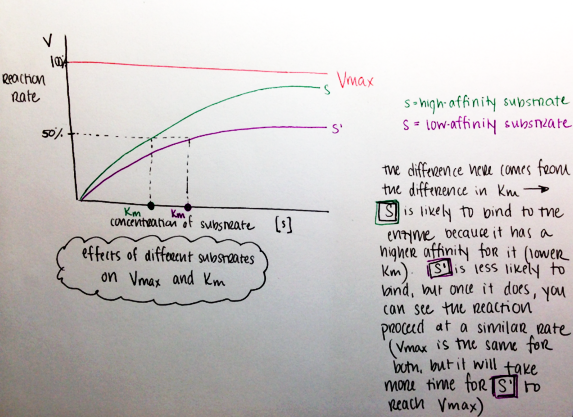
Km = dissociation constant
- Enzyme-specific
- How likely is a substrate to dissociate from an enzyme’s binding site?
- High Km values mean that dissociation is very likely; therefore, High Km = Low affinity for a substrate (if an enzyme-substrate pair are very likely to dissociate, that means the enzyme has a low affinity for, or chemical attraction to, that substrate. It may not be in chemical contact long enough for the enzyme-substrate reaction to proceed)
- Also called the Michaelis Menten constant (not discussed in this post, but Michealis Menten kinetics come with a lot of fun graphs!), and it reflects the concentration at which the reaction rate is at half its maximal value
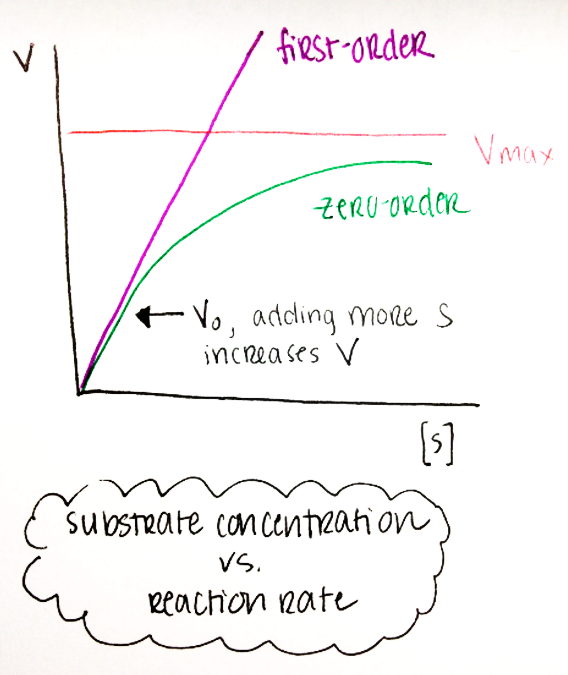
First-Order Kinetics:
When [S] is low, V0 is proportional to [S] → the amount of substrate limits the rate of the reaction (linear)
Zero-Order Kinetics:
When [S] s high, V0 reaches and asymptote (Vmax) and becomes independent of [S] → adding more substrate won’t speed up the reaction rate because the enzymes are already working as hard as they can.

Comments