
Formal charge is the charge that a bonded atom would have if its bonding electrons were shared equally. Note:
- It is not an actual charge but rather a form electron book-keeping.
- The sum of formal charges should equal the compound’s actual charge.
What formal charge tells you
There are certain instances where there is more than one feasible Lewis structure that can be drawn for a molecule. For example,
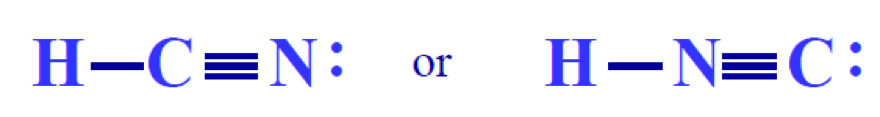
Formal charge helps us decided which structure is most likely correct. A typical general chemistry exam question may present two or more feasible Lewis structures for a molecule and ask you to pick the “best” one.
How you learned to calculate formal charge
formal charge = valence electrons – [1/2 bonding electrons + electrons in lone pairs]
OR
formal charge = valence electrons – [number of bonds + electrons in lone pairs]
Recall that the number of valence electrons for an atom is equal to its group number!
Both of the equations above will get you to the same answer. Remember that for the first equation, each bond holds two electrons; so the number of bonding electrons = 2 times the number of bonds.
Let’s use the second equation to calculate the formal charges in the molecules below:
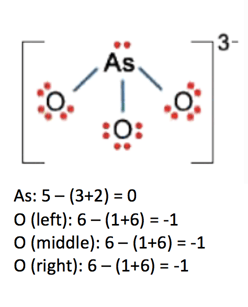
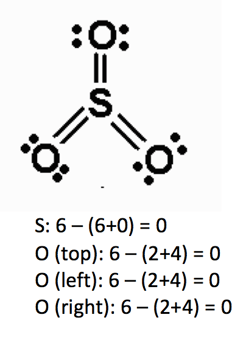
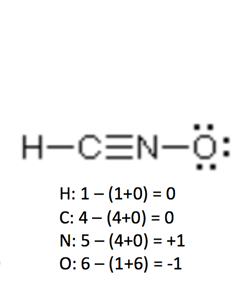
Preferred number of bonds
What it means to have a preferred number of bonds
The preferred number of bonds for an atom is exactly what it sounds like. When an atom has its preferred number of bonds AND a complete octet, its formal charge is 0, which is a good thing.
Determining the number of preferred bonds for an atom
The rules for determining the number of preferred bonds for an atom can be divided into two categories: “period 2 elements” and “period 3 and beyond elements”.
Period 2 elements
Recall that period 2 elements cannot expand their octet. They can only accommodate eight electrons which is a maximum of four bonds assuming no lone pairs. For these elements, the number of preferred bonds equals:
8 – group number
|
Atom |
H |
C |
N |
O |
F |
|
Preferred number of bonds |
1 |
4 |
3 |
2 |
1 |
Hydrogen is an exception as it can only accommodate two electrons (a duet), or one bond, irrespective of its group number.
Remember that the octet for these atoms must still be complete so lone pairs are added accordingly. For example, oxygen has a preferred bond number of 2 meaning that it will have two lone pairs. Think about the Lewis structure of water.
Period 3 and beyond elements
Recall that elements in period 3 or greater can accommodate as many as twelve electrons if it is the central atom. These elements can expand their octet if they need to. For these elements, if their octet is NOT expanded, the number of preferred bonds equals 8 – group number.
This is identical to its period 2 counterpart. Remember that members of the same group have similar chemical properties.
If the octet IS expanded, the number of preferred bonds equals: group number.
|
Atom |
Si |
P, As |
S, Se |
Cl, Br |
|
Preferred number of bonds for expanded octet (NO LONE PAIRS) |
Not applicable (4 bonds = 8 electrons, normal octet) |
5 |
6 |
Not applicable (7 bonds = 14 electrons) |
Not all elements can achieve their preferred number of bonds in the expanded octet form because only twelve electrons can be accommodated.
In addition, the element in expanded octet form cannot have any lone pairs on it or it will not achieve a formal charge of 0. This may seem intimidating to remember but it’ll click more with each practice question! Remember it as the fact that the octet is already expanded with the bonds, so no additional electrons are needed.
Determining formal charge with THe preferred number of bonds
Period 2 elements
One more bond than the preferred number:
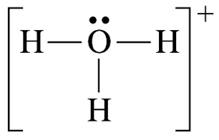
Formal charge +1
The formal charge of oxygen is +1.
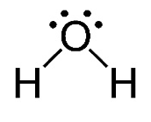
Preferred number of bonds
Formal charge = 0
The formal charge of oxygen is 0.
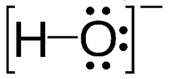
One less bond than preferred number
Formal charge = -1
The formal charge of oxygen is -1.
The rest of the trend is intuitive: two more than or two less than preferred number of bonds when applicable. Remember that period 2 elements cannot expand their octet, hence the “when applicable”.
Also remember that this approach to determining formal charge only works if octet is complete which, besides the few incomplete octet exceptions, it should be if a feasible Lewis structure was drawn. How to draw feasible Lewis structures is not covered in this post.
Period 3 and beyond elements
If the octet is not expanded, use the rules above.
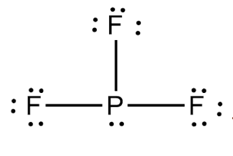
The formal charge of phosphorus is 0.
If the octet is expanded with preferred number of bonds, formal charge = 0.
The formal charge of sulfur is 0.
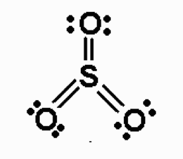
Anything other than preferred number of bonds with expanded octet, use the standard formula to determine formal charge.
Putting preferred number of bonds into practice
Let’s use preferred number of bonds to calculate the formal charge of molecules from earlier:
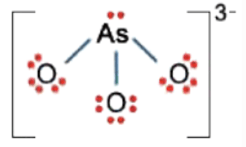
As: not expanded, preferred = 0
O (left): one less = -1
O (middle): one less = -1
O (right): one less = -1
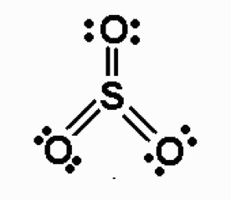
S: expanded, preferred = 0
O (top): preferred = 0
O (left): preferred = 0
O (right): preferred = 0
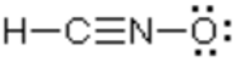
H: preferred = 0
C: preferred = 0
N: one more = +1
O: one less = -1
The same answers are achieved!
Choosing the best Lewis structure
When choosing the best Lewis structure among two or more feasible ones, remember the following rules:
- Smaller formal charges are more favorable.
- More electronegative atoms should have negative formal charges. In other words, the central atom should never have a negative formal charge if you can avoid it. In addition, if two terminal atoms are competing for a negative formal charge, the best Lewis structure will give the negative formal charge to the most electronegative of the two.
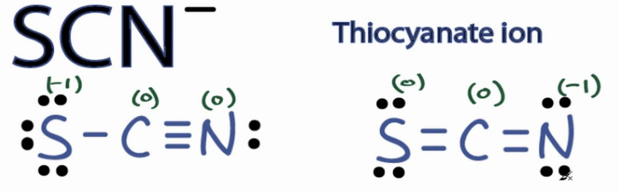
In this case, the second Lewis structure is the best because nitrogen is more electronegative than sulfur and carries the negative formal charge.
- Like charges should not be on adjacent atoms
Let’s bring back the example from the very beginning of this post. Which one is the best Lewis structure of the two?
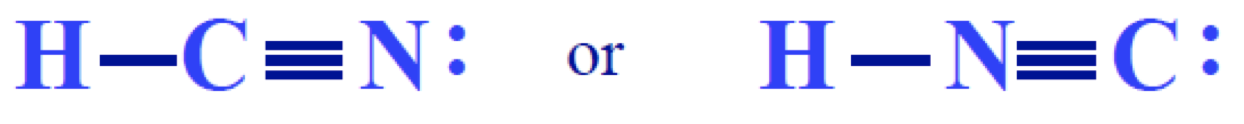
First, we must determine the formal charges for both:
H: preferred = 0 H: preferred = 0
C: preferred = 0 C: one less = -1
N: preferred = 0 N: one more = +1
The first Lewis structure is the best because there are smaller formal charges. For what it’s worth, carbon is supposed to be the central atom anyway, according to the rules of drawing Lewis structures!
Let’s do one more example for good measure. Which one is the best Lewis structure of the two?

P: not expanded, one more = +1 P: expanded, preferred = 0
O (top): one less = -1 O (top): one less = -1
O (left): one less = -1 O (left): preferred = 0
O (right): one less = -1 O (right): one less = -1
O (bottom): one less = -1 O (bottom): one less = -1
The second Lewis structure is the best because it has smaller formal charges. In addition, a negative formal charge on the central atom (phosphorus) has been avoided.
In summary, formal charges are used to help us determine the quality of a Lewis structure. Just because it’s feasible doesn’t mean it’s good. Calculating formal charges can be tedious, especially if there are numerous different atoms in the molecule. Preferred number of bonds allows to simply scan a molecule and determine the formal charges.
Comments