People often dismiss organic chemistry as “all memorization”. I disagree – organic chemistry is just a series puzzles based on a few basic concepts (electronics, sterics, orbitals) that come together to answer almost any problem you might encounter on your homework or tests.
One possible exception to this rule is retrosynthesis.
The basic idea of retrosynthesis problems is that you have a product in mind and need to figure out how to make it from basic starting materials.
Retrosynthesis problems require two major skills: (1) puzzle-solving skills and (2) a solid knowledge of reactions (which is the memorization part). Since each instructor will have his own approach towards designing these problems, I’ll offer a few general tips for students who are starting to learn retrosynthesis.
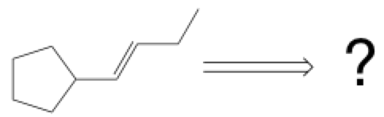
Just in case you haven’t encountered these yet, here’s an example of a retrosynthesis problem with unspecified starting materials. Note the double-lined retrosynthetic arrow which represents a backwards step.
Puzzle skills
I have to admit that puzzle-solving skills are developed only through practice. When you first try these types of problems, it will be bewildering to imagine all the possibilities. It can be particularly difficult for students who are able to do “forwards” problems easily on the first try because they are hesitant to choose a possibly incorrect path backwards and end up at a frustrating dead end. While it’s impossible to know exactly what path to take, here are some tips for improving your likelihood of developing a successful and efficient retrosynthetic plan.
- Approach these problems backwards if you are just given the final product. Don’t just guess at starting materials and try to end up at the desired product. If the starting materials and product are both specified, you can work either backwards or forwards.
- The goal of a retrosynthesis is to be as short as possible. Yes, there are a lot of different paths possible, but some will be much more efficient.
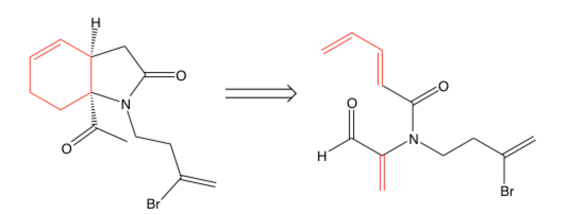
- Identify disconnection possibilities by drawing a line through bonds on the product. You will usually disconnect carbon-heteroatom bonds, but breaking apart C-C bonds is definitely possible, especially if they aren’t sp3-hybridized.
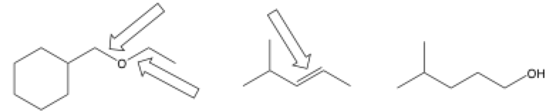
The arrows point to bonds that are likely disconnection sites. The last doesn’t have very obvious bonds to break (and could just be a starting material unless given as a product).
- The most important reactions involve conversions between alcohols, carbonyls, and halogens.
- Unless you just went over such complicated reactions or are explicitly being tested on them, avoid using more complicated reactions such as the Ruff degradation or pinacol rearrangement unless you can recognize them instantly.
- Count your carbons after every step. It’s extremely easy to lose one, especially when drawing compounds in line notation over and over. Even seasoned chemists will lose carbons once in a while.
- Check proposed backwards reactions by doing them forwards. This helps you identify any issues such as carbocation rearrangements, stereochemistry, etc. that may not lead to your product.
- Be mindful of incompatible functional groups in reactions. For example, if you want to use a Grignard reagent to react with a carboxylic acid, you need to remember that it will also react with any ketones, halides, or epoxides on the same starting material.
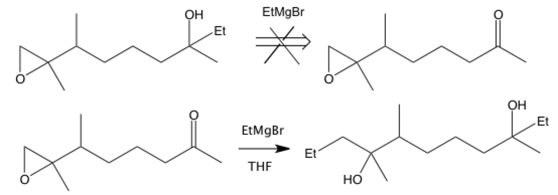
Why won’t the retrosynthetic step on the top work? It’s because adding the Grignard to the proposed reactant would result in the bottom reaction instead.
- If starting materials are unspecified, a good rule of thumb is that they should have very limited heteroatoms. If your first theoretical step involves a simple substitution (say, from a bromine to an iodine), then you should start with the second step.
- Stereochemistry matters.
Reaction Repertoire
This is where organic chemistry starts to become memorization. Technically, you could draw out each arrow-pushing step to logically determine what product would emerge from a reaction, but who has the time? It is far easier to memorize a few parameters for each reaction and use your limited exam time on other problems.
I recommend a combination of tables and graphs for memorization. You should start immediately after you start learning reactions so you can add to them incrementally instead of trying to assemble a list of forty reactions the night before the final. Update them and re-draw them when necessary. This might seem tedious, but writing repetition helps most people learn.
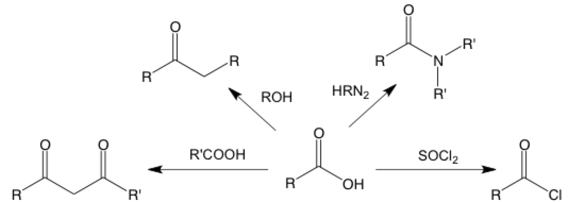
This is a simple example of a summary graph of reactions of a carboxylic acid.
Graphs should be developed for each starting functional group to illustrate the variety of products you can get in a single step. This is especially great for visual learners! Graphs are particularly useful for recognizing more complicated reactions that form carbon-carbon bonds because you will better be able to recognize the underlying structures.
A six-membered ring with a single double bond is shown in red. Ideally, you would instantly recognize this as the product of a Diels-Alder reaction and be able to draw the synthon on the right. This example comes from Raymond Funk’s synthesis of beta-erythroidine.
Tables are more complicated, but are also a good way to organize reactions. I have listed useful examples of column/rows below. At the beginning, a lot of these columns will be completely inapplicable, and that’s OK. As you learn more complicated reactions, you’ll appreciate the column header as a reminder to check.
- Reaction name. Most reactions will have a name, and it’s useful to know these. Sometimes reaction names give you clues; for example, a hydrohalogenation involves adding a hydrogen and a halogen. Many people are also better at name association than chemistry jargon, so a Williamson ether synthesis might be easier to remember.
- Reactants/products. If different starting materials undergoing the same reaction make different products, it’s useful to list the reaction in multiple rows for each possibility.
- Reagents. These are extremely important to memorize and will also act as a major clue for forward syntheses. Sometimes different reagents can achieve the same goal. Reagents might also include non-chemical things still necessary for reaction such as heat or light.
- Solvents. Sometimes they are unspecified, but some solvents are heavily associated with certain reactions so will aid in recognition. For example, Grignard reactions will usually happen in ethers. It’s also helpful to know what solvent (and reagent) abbreviations mean so you know what to do with ACN, THF, or TMSCl.
- Regiochemistry/stereochemistry/Markovnikov. Just memorizing these parameters will help you tackle retrosynthesis quickly. It’s good to also understand why reactions result in specific isomers!
- Comments. How else would you be able to remind yourself of things that don’t fit in other boxes? This is where you put fun facts such as “radical mechanism” or “not very favorable”.
Reading these tips is the easy part. Now you have to integrate them into actual problem solving. The most important tip of all in retrosyntheses (and all of chemistry) is practice, practice, practice. And if you’re still struggling, consider contacting Cambridge Coaching!

Comments