All cells are surrounded by a cell membrane that forms a barrier between the cell and its surroundings. This membrane is often referred to as the phospholipid bilayer. As you can probably tell from the name, a phospholipid bilayer is made up of two layers of lipids. The fluidity of this membrane must be maintained within a certain range for the cell to function properly. There are a number of factors that help influence membrane fluidity. Before we review those factors, let's start with a quick review of the structure of the bilayer.
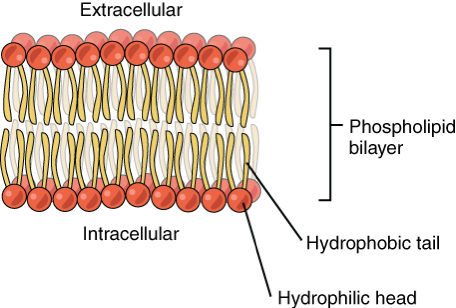
What is the phospholipid bilayer?
The phospholipid bilayer is composed of two layers of lipids. Each lipid contains a hydrophobic (water repelling) tail and a hydrophilic (water attracting) head. The lipids form into a bilayer with the hydrophobic tails facing the interior of the bilayer forming a hydrophobic region held together, in part, by intermolecular forces between the tails. The hydrophilic heads form a hydrophilic region on either side of the bilayer that can interact with the water rich environments on either side of the bilayer.
Now, let's take a look at the factors that influence membrane fluidity!
Factor #1: The length of the fatty acid tail
The length of the fatty acid tail impacts the fluidity of the membrane. This is because the intermolecular interactions between the phospholipid tails add rigidity to the membrane. As a result, the longer the phospholipid tails, the more interactions between the tails are possible and the less fluid the membrane will be.
Factor #2: Temperature
As temperature increases, so does phospholipid bilayer fluidity. At lower temperatures, phospholipids in the bilayer do not have as much kinetic energy and they cluster together more closely, increasing intermolecular interactions and decreasing membrane fluidity. At high temperatures the opposite process occurs, phospholipids have enough kinetic energy to overcome the intermolecular forces holding the membrane together, which increases membrane fluidity.
Factor #3: Cholesterol content of the bilayer
Cholesterol has a somewhat more complicated relationship with membrane fluidity. You can think of it is a buffer that helps keep membrane fluidity from getting too high or too low at high and low temperatures.
At low temperatures, phospholipids tend to cluster together, but steroids in the phospholipid bilayer fill in between the phospholipids, disrupting their intermolecular interactions and increasing fluidity.
At high temperatures, the phospholipids are further apart. In this case, cholesterol in the membrane has the opposite effect and pulls phospholipids together, increasing intermolecular forces and decreasing fluidity.
Factor #4: The degree of saturation of fatty acids tails
Phospholipid tails can be saturated or unsaturated. The terms saturated and unsaturated refer to whether or not double bonds are present between the carbons in the fatty acid tails. Saturated tails have no double bonds and as a result have straight, unkinked tails. Unsaturated tails have double bonds and, as a result, have crooked, kinked tails.
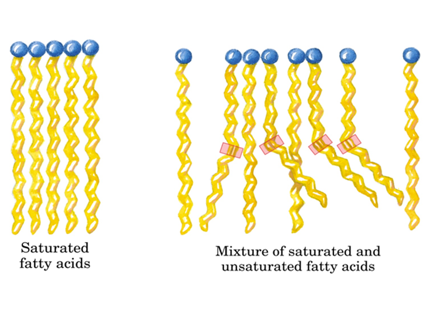
As you can see above, saturated fatty acids tails are arranged in a way that maximizes interactions between the tails. These interactions decrease bilayer fluidity. Unsaturated fatty acids, on the other hand, have more distance between the tails and thus fewer intermolecular interactions and more membrane fluidity.
In summary!

Comments