On average students scored 3.88/10 points on this question while only 1% of students got a perfect score. In this question, students are expected to know how to analyze the experimental data that is given and show an understanding of thermodynamics.
The Question
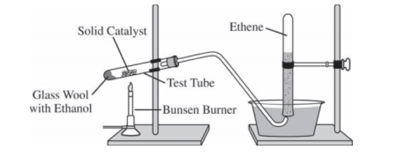
Ethene, C2H4(g) (molar mass 28.1 g/mol), may be prepared by the dehydration of ethanol, C2H5OH(g) (molar mass 46.1 g/mol), using a solid catalyst. A setup for the lab synthesis is shown in the diagram above. The equation for the dehydration reaction is given below.

A student added a 0.200 g sample of C2H5OH(l) to a test tube using the setup shown above. The student heated the test tube gently with a Bunsen burner until all of the C H OH(l) 2 5 evaporated and gas generation stopped. When the reaction stopped, the volume of collected gas was 0.0854 L at 0.822 atm and 305 K. (The vapor pressure of water at 305 K is 35.7 torr.)
(a) Calculate the number of moles of C2H4(g)
- (i) that are actually produced in the experiment and measured in the gas collection tube and
- (ii) that would be produced if the dehydration reaction went to completion.
(b) Calculate the percent yield of C2H4(g) in the experiment.
Because the dehydration reaction is not observed to occur at 298 K, the student claims that the reaction has an equilibrium constant less than 1.00 at 298 K.
(c) Do the thermodynamic data for the reaction support the student’s claim? Justify your answer, including a calculation of DG298 D for the reaction.
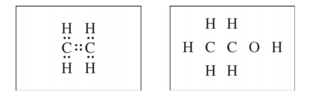
(d) The Lewis electron-dot diagram for C2H4 is shown below in the box on the left. In the box on the right, complete the Lewis electron-dot diagram for C2H5OH by drawing in all of the electron pairs.
(e) What is the approximate value of the C–O–H bond angle in the ethanol molecule?
(f) During the dehydration experiment, C2H4(g) and unreacted C2H5OH(g) passed through the tube into the water. The C2H4 was quantitatively collected as a gas, but the unreacted C2H5OH was not. Explain this observation in terms of the intermolecular forces between water and each of the two gases.
The Solution
First, quickly read through the question and get an idea of on what you are being tested. Underline values that are given! Notice that both the standard enthalpy change of reaction(ΔH°298) and standard entropy of reaction (ΔS°298) are given in different units. They are also given at standard temperature and pressure. During the test make sure to convert all measurements into the correct units before doing calculations and get in the habit of writing out the units in the answer!
Part (a)
Now, let’s work through (a) which asks for (i) the moles actually produced in the experiment (ie. experimental yield) and (ii) the moles that would be produced if the reaction went to completion (ie. theoretical yield). Before hand, remember to clearly label your work and answers for section (a)(i) vs (a)(ii). Points are not given for correct answers in the wrong section. Recognize that we need the equation PV=nRT from the equation table where n represents the moles and to solve for moles the equation becomes n=PV/RT. The question gives us the volume of gas collected, 0.0854L, total pressure, 0.822 atm, and temperature, 305K. Notice that the total pressure is given but we need the pressure of ethene. Given the equation Ptotal = Pwater + Pethene from the equation table we can solve for Pethene. However, we first need to convert Pwater into the correct units, atm. Given Pwater = 35.7 torr and 1atm = 760 torr using division we can cancel the units and get Pwater = 0.0470atm. Remember the significant figures here. Solving for Pethene = Ptotal – Pwater = 0.822atm – 0.0470 atm = 0.775atm. Using this pressure and the equation n=PV/RT = (0.775 atm)(0.0854 L)/ (0.08206 L atm mol-1 K-1) ( 305 K) = 0.00264 mol. Make sure to write in all the units and make sure they cancel out to leave moles! For (a)(ii), which asks for the theoretical yield of moles if the reaction went to completion make sure the equation is balanced. Since in this case the equation is already balanced we see that 1mol C2H5OH will yield 1 mol of C2H4 thus calculate the moles of C2H5OH given the amount of grams initially used.
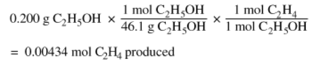
Part (b)
Part (b) wants you to use the experimental yield and theoretical yield and calculate the percent yield. Many students calculated the percent error here but remember that the percent yield = actual yield/theoretical yield x 100. Percent error is not the same as percent yield so be careful! Using this equation 0.00264 mol/0.00434 mol x 100 = 60.8%. Since we are representing the percent yield there is no need to convert moles to grams. Also, to check your answer make sure that the percent yield is not more than 100% since it doesn’t make sense to yield more than the theoretical amount possible.
Part(c)
Part (c) requires that you know how to calculate ΔG° of the reaction using the equation ΔG°= ΔH°-TΔS° that is given on the equation sheet and determine the equilibrium constant for gas pressure, Kp, from this equation. First let’s calculate ΔG° at 298K like the question requires. Note that ΔH° and ΔS° are already given at 298K but in different units. Convert the units of ΔS° to kJ by diving by 1000 since 1J = 1000kJ. Thus,

The student claims the reaction has an equilibrium constant less than 1.00 at 298K. To determine whether this is true let’s use the equation give from the equation sheet that includes both ΔG° and Kp, which is ΔG°= -RT ln Kp. Solving for Kp we get Kp= e(-ΔG°/RT) < 1. So, yes the data supports the students claim because a ΔG° > 0 results in a Kp < 1.
Part (d)
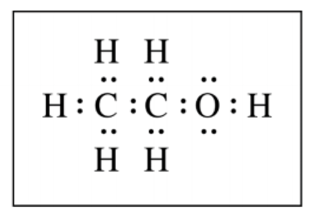
Part (d) can be answered without any calculation. For (d) you are expected to know how to draw Lewis structures which are based on valence electrons. Remember that for neutral atoms the group number is equal to the number of valence electrons. Thus Carbon has 4 valence electrons, Oxygen has 6, and Hydrogen has 1 valence electron, which gives 20 valence electrons that need to be represented in the dot structure. Begin by drawing two dots between each atom to represent a chemical bond. By doing this you will use 16 valence electrons and satisfy the octet rule for both carbons but 4 electrons are left and the octet for oxygen is not satisfied. Thus those 4 electrons go around the oxygen and represent two lone pairs, so the diagram looks like this:
 Part (e)
Part (e)
Part (e) asks you to give the bond angle between the C-O-H in this molecule. Due to the lone pairs present around the oxygen this bond makes a tetrahedral like angle, which is approximately 109°. However, the scorers gave 1 point for any bond angle between 100° - 115°.
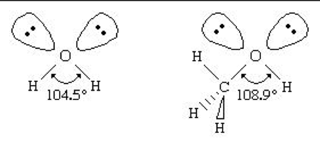
This question required knowledge about VSEPR models but required no justification to receive points.
Part (f)
This part wants you to show an understanding of the intermolecular forces between ethene and water and ethanol and water. Ethene, which is a nonpolar molecule, is only slightly soluble in water because of the weak dipole/induced dipole forces of nonpolar molecules. Ethanol, however, is soluble in water because they are polar and form hydrogen bonds with the water molecules as they dissolve.
Using this question as a study guide I would advise you to review the following:
- VSEPR bond angles
- types of reactions (ie. endothermic and exothermic)
- relationship between enthalpy, entropy, and Gibbs free energy
- intramolecular forces and physical properties
- ideal gas law
Always remember to show your work, label each part of the question correctly, use significant figures, and watch the units!
Comments