Determining which of two molecules is more acidic is tricky if you haven’t yet organized those factors that influence acidity. The protocol is a method I learned from my mastermind Orgo 2 professor to keep these ideas in order when they come into conflict. Namely:
Size is more important than
Electronegativity, which is more important than
Resonance, which trumps the
Inductive Effect.
How to Use the Protocol
When determining acidity, first make the change (deprotonate the atom of interest), and then evaluate the change. As we know, the stability of the resultant negative charge dictates the original molecule’s acidity. Anything that stabilizes this new negative charge makes the original acid more acidic (i.e. lower pKa, higher Ka). Anything that destabilizes the negative charge makes the original acid less acidic (i.e. higher pKa, lower Ka).
This makes sense, right? When we’re deprotonating an acid we’re bringing the acid to an unhappy place: a place with a full-blown negative charge. (We like neutrality–not charge!) Of course, then, in picking the more acidic molecule we should pick the one being brought to the better of the two places. In other words, minimize the discomfort!
When comparing basicity, we want to ask ourselves which of the two bases we’re looking at is more comfortable with its electrons. Bases that are more comfortable with their electrons are less basic.
This also makes sense. If I’m comfortable with my electrons, why would I give them away to a proton?
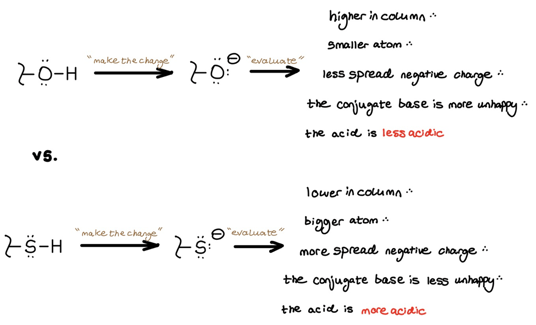
1. Size
We use size when comparing atoms in the same column of the periodic table. Atoms lower in the periodic table are larger and better able to spread out and stabilize negative charges. This explains why thiols are more acidic than alcohols and why acid halides become more acidic as you move down the column of halogens.
2. Electronegativity
We use electronegativity when comparing atoms in the same row of the periodic table. Atoms to the right of the periodic table are more electronegative and therefore better able to withstand a negative charge. This explains why alcohols are more acidic than amines.
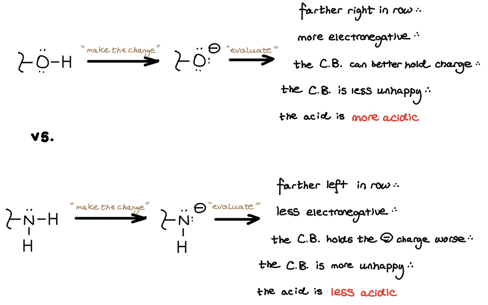
What about basicity? Why is Orgo’s hallmark basic functional group the amine and not the alcohol? Oxygen is to the right of and more electronegative than nitrogen. Therefore, oxygen holds onto its lone pair electrons tighter and is more comfortable hanging onto them. An amine’s nitrogen holds onto its lone pair looser and is less comfortable hanging onto them.
Side Note: electronegativity also comes into play when comparing two of the same atom with different hybridizations. For example, a sp3 hybridized nitrogen is less electronegative than a sp2 hybridized nitrogen, which is less electronegative than a sp hybridized nitrogen. Think about how this affects basicity.
3. Resonance
If you’ve made it this far down the protocol, it’s because you’re comparing charges on the same atom (with identical size and electronegativity). Your new job is to count resonance options. The conjugate base with more resonance options has more ways to delocalize and stabilize the resultant negative charge. As always, minimize the discomfort!
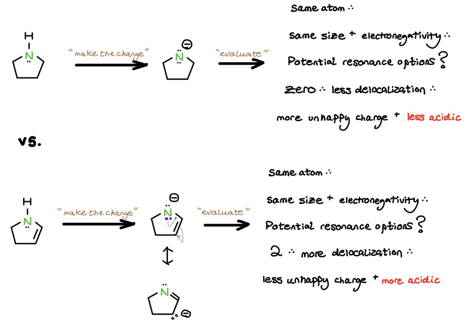
What about basicity? A negatively-charged base with more resonance options is clearly more comfortable with its electrons than a base with fewer resonance options. This makes negatively-charged bases with additional delocalization less basic!
4. Inductive Effect
You made it this far down the protocol if you didn’t find resonance options in either molecule or if you’re comparing two carboxylic acids. (Carboxylates have two and only two resonance options, which gives us a tie at the protocol’s resonance level.)
Recall that inductive donors are general alkyl groups (e.g. methyls and ethyls). After making the change and evaluating, we see that alkyl inductive donors destabilize negative conjugate bases. I tell my students that such donors blow up the negative charge.
Conversely, inductive withdrawers like halogens stabilize negative charges. I tell my students that such withdrawers quench the negative charge.
What about basicity? If you’re a base with a blown-up negative charge, you’re quite uncomfortable with your electrons and much more apt to throw them at a proton. On the other hand, if you’re a base with a quenched negative charge, you’re in a more comfortable position and less apt to give away your electrons.
Conclusion
The last thing you should do in orgo is memorize statements like “more resonance = more acidic.” Statements like these are meaningless because they don’t rely on a string of logic. The protocol gives you the theory necessary to reason through your acid-base problems without having to rely on tables of pKa values–my absolute biggest pet peeve! Use your head, and don’t forget:

Comments